Biogen’s Litifilimab Earns FDA Breakthrough Therapy Designation for Cutaneous Lupus Erythematosus—First Targeted Therapy on the Horizon
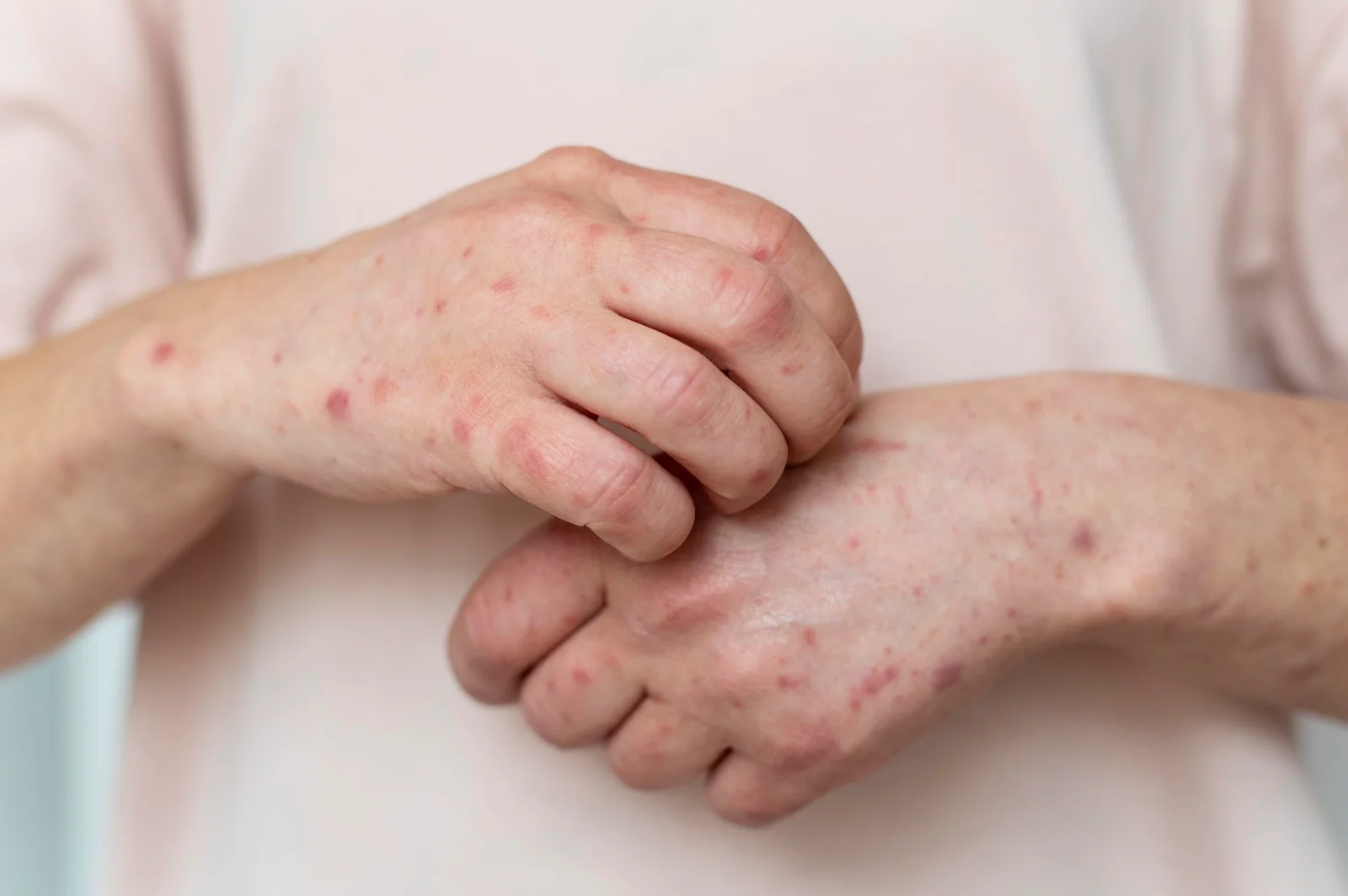
Biogen Inc. announced today that the U.S. Food and Drug Administration (FDA) has granted Breakthrough Therapy Designation for litifilimab (BIIB059) for the treatment of cutaneous lupus erythematosus (CLE). Litifilimab is a first in-class, humanized IgG1 monoclonal antibody (mAb) targeting blood dendritic cell antigen 2 (BDCA2). CLE is a chronic autoimmune disease affecting the skin that currently has no targeted treatments.
“The breakthrough therapy designation for litifilimab illustrates the FDA’s recognition of cutaneous lupus as a serious disease that urgently requires new therapies,” said Victoria Werth, MD, MS, a professor of Dermatology at the Perelman School of Medicine at the University of Pennsylvania, and one of the researchers who is conducting the phase 3 trial. “With topical steroids and antimalarials as the initial therapies for managing CLE and no alternatives specifically approved for CLE, there is a need for effective, targeted treatments, and that could be a drug like litifilimab.”
The designation is intended to expedite the development and review of drugs for serious conditions, and is based on the totality of litifilimab data, including the results from the Phase 2 LILAC study. The LILAC data were previously published in The New England Journal of Medicine and demonstrated that litifilimab reduced skin disease activity in people with CLE compared to placebo. The current standard of care for CLE includes topical steroids, antimalarials and immunosuppressants. While current treatments help manage symptoms, they do not alter the progression of the disease.
“The FDA grants breakthrough therapy designation to programs based on the seriousness of the condition and the potential of the therapeutic candidate to provide substantial improvements over available therapies. The FDA’s designation reinforces Biogen’s belief that litifilimab could be a first-in-class therapy targeting BDCA2 for cutaneous lupus erythematosus,” said Priya Singhal, M.D., M.P.H., Executive Vice President and Head of Development at Biogen. “This designation is a significant milestone for litifilimab as we advance the ongoing AMETHYST Phase 3 study, with the goal of bringing a new potential therapeutic option to the millions of people living with CLE.”
Biogen is continuing to evaluate the efficacy and safety of litifilimab in the AMETHYST Phase 3 study, with a data readout expected in 2027. More information on the AMETHYST study (NCT05531565) is available at clinicaltrials.gov and BiogenTrialLink.
“The Lupus Research Alliance is dedicated to advancing lupus research, and today’s FDA Breakthrough Therapy designation for litifilimab reinforces our shared understanding of cutaneous lupus as a serious, debilitating condition that urgently needs therapies that can alter the course of the disease,” said Albert T. Roy, President & CEO of the Lupus Research Alliance.
“Incorporating the voices of people living with cutaneous lupus is vital to advancing drug development, and through our clinical affiliate, Lupus Therapeutics, we are proud to collaborate with Biogen on the cutaneous lupus erythematosus clinical trials for litifilimab. As a convenor bringing together leading industry partners, clinicians, patients, and FDA experts, the Lupus Research Alliance is encouraged by this progress to accelerate a potential new treatment that may improve the quality of life for those affected by CLE.”
About Litifilimab (BIIB059)
Litifilimab (known as BIIB059), discovered and developed in-house by Biogen scientists, is a humanized IgG1 monoclonal antibody (mAb) targeting BDCA2 and is being investigated for the potential treatment of systemic lupus erythematosus (SLE) and cutaneous lupus erythematosus (CLE). BDCA2 is a receptor that is predominantly expressed on a subset of human immune cells called Plasmacytoid Dendritic Cells (pDCs). Binding of litifilimab to BDCA2 has been shown to reduce production of pro-inflammatory molecules by pDCs, including type-I interferon (IFN-I) as well as other cytokines and chemokines.1,2
These pro-inflammatory mediators are thought to play a major role in the pathogenesis of systemic and cutaneous lupus. Litifilimab is an investigational therapeutic candidate that has not yet been approved by any regulatory authority and its safety and effectiveness have not been established.
About Cutaneous Lupus Erythematosus (CLE)
CLE, a type of lupus, is a chronic autoimmune skin disease that can occur with or without systemic manifestations; people with CLE frequently experience symptoms including rash, pain, itch and photosensitivity as well as skin damage that may worsen over time and can include irreversible scarring, alopecia and dyspigmentation that can be disfiguring and substantially impact quality of life.3-6
Although anyone can develop lupus, an estimated 90 percent of people living with lupus are women; most begin to see symptoms between the ages of 15-40.7 The disease disproportionately impacts diverse ethno-racial groups, including African American, Asian, American Indian/Alaskan Native and Hispanic/Latino communities.8-10 There is currently no cure for lupus.
About Biogen
Founded in 1978, Biogen is a leading biotechnology company that pioneers innovative science to deliver new medicines to transform patients’ lives and to create value for shareholders and our communities. We apply deep understanding of human biology and leverage different modalities to advance first-in-class treatments or therapies that deliver superior outcomes. Our approach is to take bold risks, balanced with return on investment to deliver long-term growth.
Source Link: https://investors.biogen.com/




