Regulatory Milestone Enables European Expansion of Advanced Proton Therapy
Why is proton therapy still out of reach for many European cancer centers? Mevion Medical Systems, the global leader in compact proton therapy, has announced that the MEVION S250-FIT Proton Therapy System has received CE Marking under Regulation (EU) 2017/745 (EU MDR). This regulatory approval paves the way for the marketing and clinical use of the MEVION S250-FIT across the European Union, building on its existing U.S. FDA 510(k) clearance granted in September 2025.
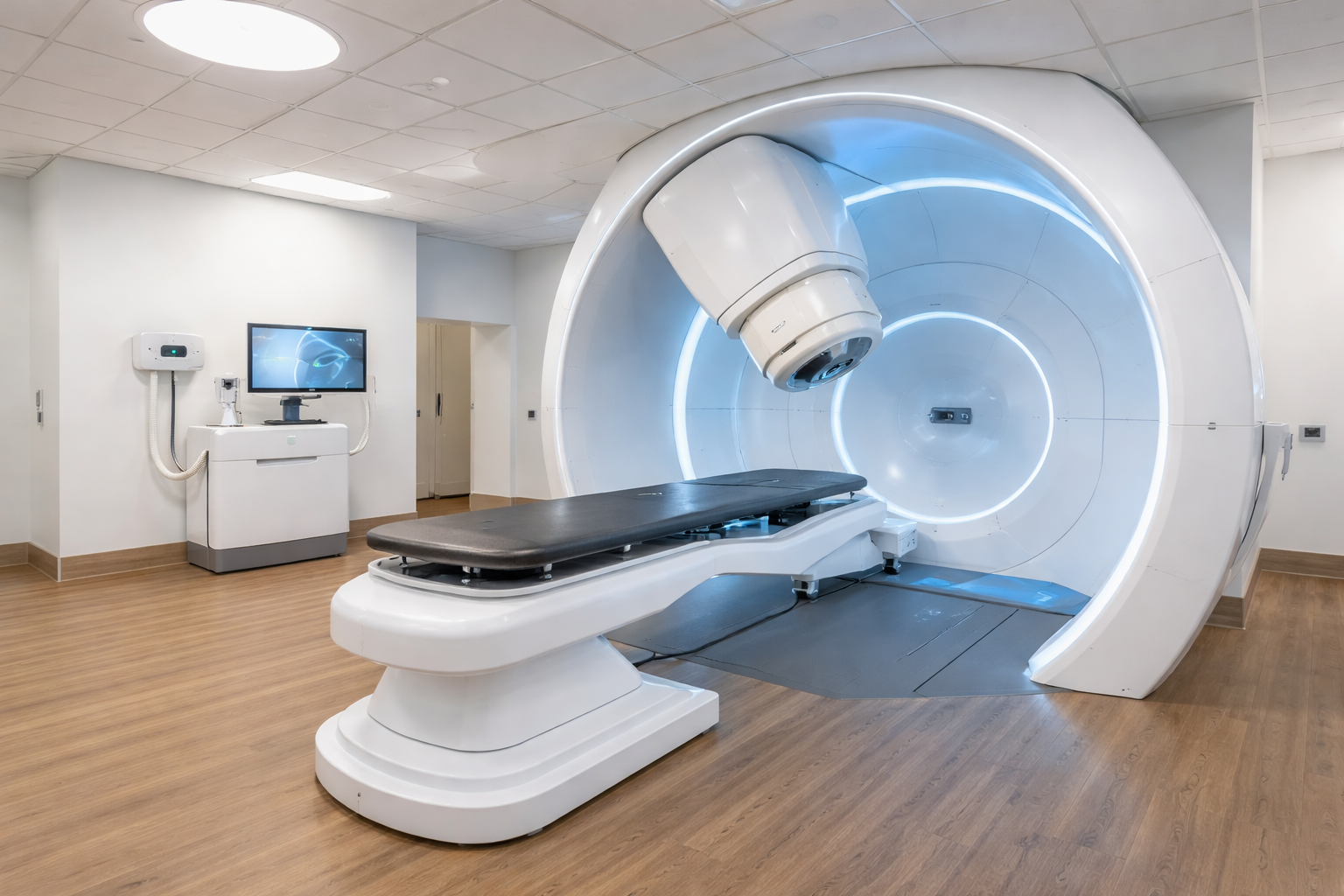
The MEVION S250-FIT is the first and only proton therapy system designed to fit into a standard radiation therapy vault, significantly reducing the cost, complexity, and timeline associated with proton therapy adoption. This breakthrough opens new pathways for hospitals and cancer centers to bring advanced proton treatment to their patients.
Key Insights at a Glance
- CE Marking: The MEVION S250-FIT is now approved for marketing and clinical use in the European Union.
- Cost Reduction: The system fits into existing radiation therapy vaults, eliminating the need for purpose-built bunkers.
- Advanced Capabilities: Features HYPERSCAN® pencil beam scanning and DirectARC™ proton arc therapy.
- Global Momentum: Mevion has signed contracts with nine leading institutions globally.
Why Infrastructure Barriers Have Hindered Proton Therapy Adoption
Proton therapy has long been recognized for its clinical benefits, but the significant infrastructure and financial barriers have limited its adoption in many European health systems. Traditional proton therapy systems require purpose-built bunkers, extensive construction, and multi-year capital projects, making them inaccessible to many hospitals and cancer centers. This has left many patients without access to this advanced form of cancer treatment, despite its proven advantages in reducing side effects and improving outcomes.
The Regulatory Clock Is Ticking for European Health Systems
Just as a sprinter must time their final push to cross the finish line, Mevion Medical Systems is racing to bring the MEVION S250-FIT to European health systems before the regulatory window closes. The CE Marking under Regulation (EU) 2017/745 (EU MDR) is a critical milestone that enables the marketing and clinical use of the system across the European Union. This timing is crucial as health systems look to modernize their cancer treatment offerings and meet the growing demand for advanced therapies.
Mevion Medical Systems Pioneers Accessible Proton Therapy
Mevion Medical Systems is revolutionizing the way proton therapy is delivered by addressing the infrastructure and financial barriers that have hindered its adoption. The MEVION S250-FIT Proton Therapy System is designed to fit into standard radiation therapy vaults, dramatically reducing the cost, complexity, and timeline traditionally associated with proton therapy. This innovation opens new pathways for hospitals and cancer centers to bring advanced proton treatment to their patients.
The MEVION S250-FIT features HYPERSCAN® pencil beam scanning for Intensity Modulated Proton Therapy (IMPT), delivering highly conformal dose distributions with hyper-fast energy layer switching and the Adaptive Aperture® proton multi-leaf collimator (pMLC). The system also supports DirectARC™ proton arc therapy, enabling VMAT-like speed and efficiency in proton treatment delivery. Additionally, it incorporates a large-bore diagnostic CT aligned to the isocenter, enhancing image-guided radiation therapy (IGRT) and adaptive treatment workflows.
“With both FDA clearance and CE Marking now in hand, the MEVION S250-FIT is positioned to transform how proton therapy is delivered worldwide,” said Tina Yu, Ph.D., CEO and President of Mevion Medical Systems. “For European health systems that have long recognized the clinical benefits of proton therapy but faced significant infrastructure and financial barriers, the S250-FIT offers a fundamentally new approach, one that brings high-quality proton therapy directly into existing treatment environments.”
Future Outlook
The CE Marking is a defining moment for accessible cancer care in Europe and beyond. As Mevion continues to build on its global momentum, with contracts signed with nine leading institutions and installations at major medical centers, the future of proton therapy looks promising. The company’s commitment to advancing the design and accessibility of proton therapy worldwide is set to bring high-quality treatment to more patients, enhancing clinical outcomes and patient satisfaction.
Conclusion
The CE Marking of the MEVION S250-FIT Proton Therapy System marks a significant step forward in making advanced proton therapy accessible to European health systems. For cancer centers and hospitals, this means a practical and cost-effective pathway to establishing or expanding proton therapy programs. How is your institution preparing to leverage this new technology? Join the conversation in the comments below.
Source link: https://www.businesswire.com/