Biogen Launches Phase 3 Trial of Felzartamab for Primary Membranous Nephropathy
Biogen Launches Global Phase 3 PROMINENT Trial Evaluating Felzartamab in Primary Membranous Nephropathy Biogen Inc. (Nasdaq: BIIB) has announced the initiation of dosing in its global Phase 3 clinical trial,…
Takeda’s GAMMAGARD LIQUID ERC Approved by FDA as First Ready-to-Use Low-IgA Immunoglobulin Therapy
Takeda Secures FDA Approval for GAMMAGARD LIQUID ERC, the First Ready-to-Use Liquid Immunoglobulin Therapy with Low IgA Content Takeda (TSE:4502/NYSE:TAK) announced today that the U.S. Food and Drug Administration (FDA)…

Merck Completes SpringWorks Acquisition to Boost Healthcare Growth
Merck Finalizes $3.4 Billion Acquisition of SpringWorks Therapeutics to Strengthen Rare Tumor Portfolio and Drive Sustainable Healthcare Growth Merck, a global leader in science and technology, today announced the successful…
Vertex’s ALYFTREK® Approved in EU for Cystic Fibrosis Treatment
Vertex Gains EU Approval for ALYFTREK®, a Next-Generation CFTR Modulator for Cystic Fibrosis Patients Vertex Pharmaceuticals (Nasdaq: VRTX) announced today that the European Commission has granted marketing authorization for ALYFTREK®…

Riliprubart Receives Orphan Drug Designation in Japan for CIDP
Riliprubart Receives Orphan Drug Designation in Japan for Chronic Inflammatory Demyelinating Polyneuropathy Sanofi announced that Japan’s Ministry of Health, Labour and Welfare (MHLW) has granted orphan drug designation to riliprubart,…

Amgen Reports Positive Phase 3 Results for Bemarituzumab in FGFR2b+ Gastric Cancer
Amgen Reports Positive Phase 3 Results for Bemarituzumab in FGFR2b+ Gastric Cancer, Marking Milestone in Targeted Therapy Amgen (NASDAQ:AMGN) has announced a significant breakthrough in the treatment of gastric and…

Strategic Leadership Changes Announced by Roche
Roche Announces Key Leadership Transitions Within Enlarged Corporate Executive Committee Roche (SIX: RO, ROG; OTCQX: RHHBY) today announced significant changes to its leadership team, as two esteemed members of its…
AbbVie to Acquire Capstan to Boost Immunology Innovation
AbbVie to Acquire Capstan Therapeutics to Advance In Vivo CAR-T for Autoimmune Diseases AbbVie and Capstan Therapeutics have announced a definitive agreement for AbbVie to acquire Capstan in a move…
FDA Reviews Allergan’s SKINVIVE PMA for Neck Appearance Improvement
FDA Accepts Allergan’s Application to Expand SKINVIVE by JUVÉDERM® for Neck Line Treatment Allergan Aesthetics, an AbbVie company (NYSE: ABBV), announced that the U.S. Food and Drug Administration (FDA) has…
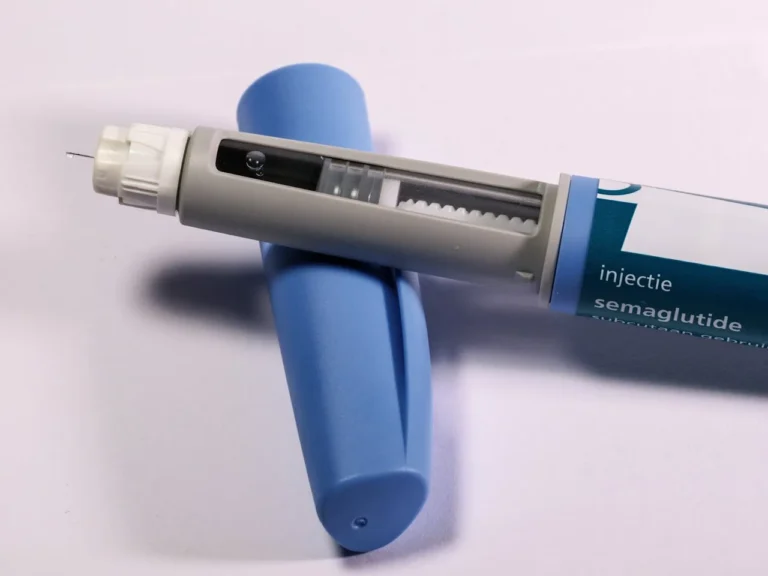
Evaluating the Risk of NAION in Patients Using Semaglutide Medications
WHO Issues Global Safety Alert Over Vision Loss Risk Linked to Semaglutide-Based Medications The World Health Organization (WHO) has issued a global safety communication directed at healthcare professionals and regulatory…
WHO Scientific Group Publishes Findings on COVID-19’s Origins
WHO Scientific Advisory Group Releases In-Depth Report on the Origins of COVID-19 Amid Ongoing Calls for Transparency and Global Cooperation The World Health Organization (WHO) has taken another step forward…

WHO and Key Stakeholders Launch Disability Health Equity Partnership
WHO Launches Landmark Global Initiative to Advance Health Equity for Over 1.3 Billion People with Disabilities In a pivotal step toward realizing global health equity, the World Health Organization (WHO)…


