
Sanofi and Alnylam Secure Landmark RNAi Approval for Hemophilia A & B
Sanofi and Alnylam Secure Landmark RNAi Approval for Hemophilia A & B Sanofi and Alnylam have achieved a significant milestone with the U.S. Food and Drug Administration (FDA) granting approval…

European Commission Approves TIVDAK® for Recurrent or Metastatic Cervical Cancer
European Commission Approves TIVDAK® for Recurrent or Metastatic Cervical Cancer Genmab A/S has announced that the European Commission (EC) has officially granted marketing authorization for TIVDAK® (tisotumab vedotin), an antibody-drug…

BeiGene’s TEVIMBRA® Gets CHMP Nod for First-Line Lung Cancer Treatment
BeiGene’s TEVIMBRA® Gets CHMP Nod for First-Line Lung Cancer Treatment BeiGene, Ltd., a global oncology company soon to be rebranded as BeOne Medicines Ltd., has announced a significant milestone in…

WuXi Biologics Achieves ISO 22301 Certification and Expands ISO/IEC 27001 Compliance
WuXi Biologics Achieves ISO 22301 Certification and Expands ISO/IEC 27001 Compliance WuXi Biologics (“WuXi Bio”) (2269.HK), a globally recognized Contract Research, Development, and Manufacturing Organization (CRDMO), has announced a significant…

Ono & Reborna Partner for CNS RNA-Targeting Drug Discovery
Ono & Reborna Partner for CNS RNA-Targeting Drug Discovery Ono Pharmaceutical Co., Ltd. (Headquartered in Osaka, Japan; President and COO: Toichi Takino; hereafter referred to as “Ono”) has announced a…

Guideline Development Group Maternal Peripartum Infection Treatment
Maternal sepsis is a life-threatening condition that arises from an infection during pregnancy, childbirth, post-abortion, or the postpartum period. It is a significant public health challenge and ranks as the…
WHO Experts Plan Bold Steps to Strengthen Global Immunization
WHO Experts Plan Bold Steps to Strengthen Global Immunization The World Health Organization’s Strategic Advisory Group of Experts on Immunization (SAGE) has raised significant concerns about mounting pressures that threaten…

Halozyme Announces Positive CHMP Opinion for Bristol Myers Squibb’s Subcutaneous Opdivo® with ENHANZE
Halozyme Announces Positive CHMP Opinion for Bristol Myers Squibb’s Subcutaneous Opdivo® with ENHANZE Halozyme Therapeutics, Inc. (NASDAQ: HALO) recently announced a significant milestone in oncology treatment development, as Bristol Myers…
ACC 2025 Sensydia CPS™ Accurately Assesses Mean Pulmonary Artery Pressure Non-Invasively
ACC 2025 Sensydia CPS™ Accurately Assesses Mean Pulmonary Artery Pressure Non-Invasively Sensydia, a clinical-stage company specializing in non-invasive cardiac assessment technologies, has announced promising results from a recent clinical study…

Novotech Secures Investment from GIC, Temasek, and TPG to Drive Global Expansion
Novotech Secures Investment from GIC, Temasek, and TPG to Drive Global Expansion Novotech, a globally recognized full-service biotech specialist clinical research organization (CRO), has announced a significant investment from leading…
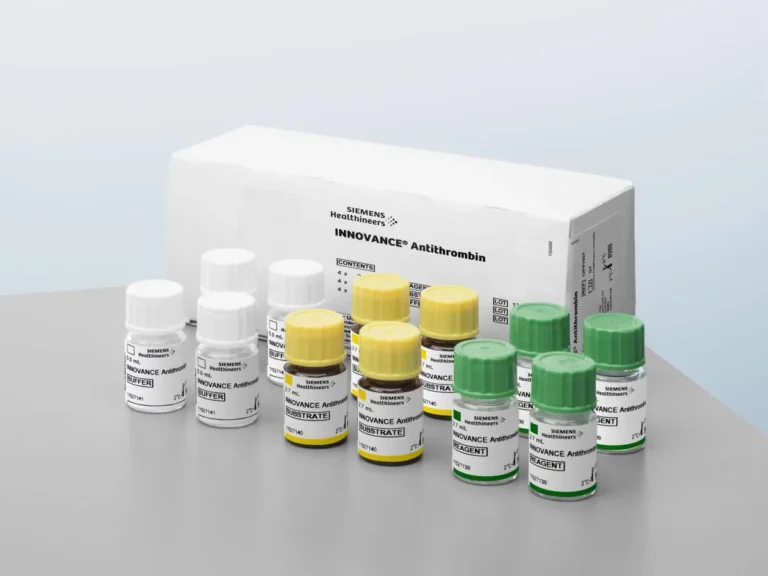
FDA Clears Siemens Antithrombin Test as Companion Diagnostic for Qfitlia
FDA Clears Siemens Antithrombin Test as Companion Diagnostic for Qfitlia Siemens Healthineers has announced that its Innovance Antithrombin assay has received FDA clearance for an additional claim, allowing it to…

BMS Gets CHMP Nod for Opdivo-Based Perioperative Regimen in Resectable NSCLC
BMS Gets CHMP Nod for Opdivo-Based Perioperative Regimen in Resectable NSCLC Bristol Myers Squibb has announced that the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines…

