
ICON Issues Financial Guidance for Full Year 2024
ICON plc, (NASDAQ: ICLR), a world-leading clinical research organisation powered by healthcare intelligence, today announced its financial guidance for the year ending December 31, 2024. For the full year 2024, revenue is expected…

Biotech companies to increase R&D spend but highlight complexity of clinical trials – ICON
ICON plc, (NASDAQ: ICLR) a world-leading healthcare intelligence and clinical research organisation, today launched its “Optimising biotech funding” whitepaper. This provides an overview of the current state of play for biotech…

ICON wins industry accolades from TIME Magazine, Forbes and Financial Times in second half of 2023
ICON plc, (NASDAQ: ICLR) a world-leading healthcare intelligence and clinical research organisation (CRO), finishes the second half of 2023 with positive recognition as a top-rated employer and leader in clinical trial…

Bayer’s AskBio initiates Phase II Gene PHIT trial in Congestive Heart Failure (CHF)
Bayer AG and Asklepios BioPharmaceutical, Inc. (AskBio), a gene therapy company wholly owned and independently operated as a subsidiary of Bayer AG, have announced the initiation of GenePHIT (Gene PHosphatase…

Bayer announces key leadership changes for its Crop Science division
Bayer is applying several key changes to the Executive Leadership Team of its Crop Science Division. The new setup will support the company to shape agriculture for farmers, consumers, and…
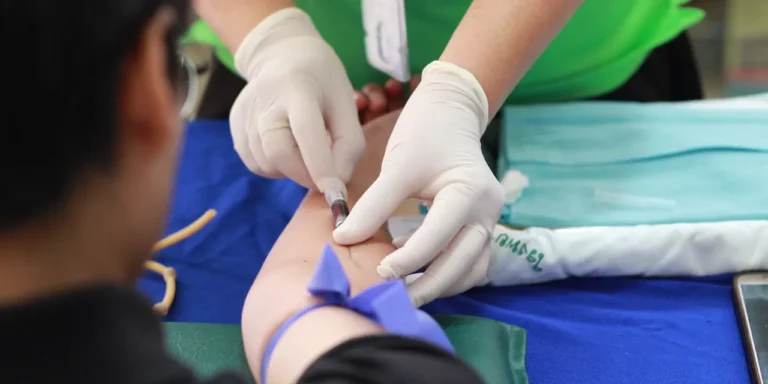
“LEQEMBI®” Approved for the Treatment of Alzheimer’s Disease in China
Eisai Co., Ltd. (Headquarters: Tokyo, CEO: Haruo Naito, “Eisai”) and Biogen Inc. (Nasdaq: BIIB, Corporate headquarters: Cambridge, Massachusetts, CEO: Christopher A. Viehbacher, “Biogen”) announced today that humanized anti- soluble aggregated…

Nucala approved in China for use in severe asthma with an eosinophilic phenotype
GSK plc (LSE/NYSE: GSK) today announced that the China National Medical Products Administration has approved Nucala (mepolizumab), as an add-on maintenance treatment for severe eosinophilic asthma in adults and adolescents aged 12…

GSK enters agreement to acquire Aiolos Bio
GSK plc (LSE/NYSE: GSK) and Aiolos Bio, Inc. (Aiolos) today announced that they have entered into an agreement under which GSK will acquire Aiolos, a clinical-stage biopharmaceutical company focused on…

Aetna® helps treat childhood vision disorders with $75,000 donation to Plano Child Development Center
Aetna Better Health of Illinois®, a CVS Health® company (NYSE: CVS), announced that Aetna® has provided a community investment of $75,000 to Plano Childhood Development Center to help children receive the…

Vertex Announces Approval of First CRISPR/Cas9 Gene-Edited Therapy, CASGEVY™, for the Treatment of Sickle Cell Disease and Transfusion-Dependent Beta Thalassemia in Kingdom of Saudi Arabia
Vertex Pharmaceuticals Incorporated (Nasdaq: VRTX) announced today that the Saudi Food and Drug Authority (SFDA) granted Marketing Authorization for CASGEVY™ (exagamglogene autotemcel [exa-cel]), a CRISPR/Cas9 gene-edited therapy, for the treatment of sickle cell…

Pfizer Recommends Shareholders Reject the Below-Market Mini-Tender Offer by TRC Capital Investment Corporation
Pfizer Inc. (NYSE: PFE) today announced that it has received notice of an unsolicited mini-tender offer by TRC Capital Investment Corporation of Ontario, Canada to purchase up to 4 million…

TIVDAK® Supplemental Biologics License Application Accepted for Priority Review by FDA for Patients with Recurrent or Metastatic Cervical Cancer
Pfizer Inc. (NYSE: PFE) and Genmab A/S (Nasdaq: GMAB) announced today that the U.S. Food and Drug Administration (FDA) has accepted the supplemental Biologics License Application (sBLA) seeking to convert the accelerated approval…

