LEO Pharma Presents New Long-Term Data for SPEVIGO® (spesolimab-sbzo) in Adults with Generalized Pustular Psoriasis at AAD 2026
LEO Pharma A/S has announced new long-term clinical data supporting the use of SPEVIGO® (spesolimab-sbzo) in patients with generalized pustular psoriasis (GPP), a rare and severe inflammatory skin disorder. The findings, derived from the ongoing EFFISAYIL® ON open-label extension study, were presented at the American Academy of Dermatology Annual Meeting, highlighting the therapy’s sustained efficacy, safety, and tolerability over extended treatment durations.
The EFFISAYIL ON study builds on the pivotal EFFISAYIL 1 and EFFISAYIL 2 trials and is designed as a five-year extension to evaluate long-term outcomes in patients receiving SPEVIGO. This extended evaluation is particularly important in the context of GPP, a chronic, systemic, and often unpredictable disease characterized by recurrent flares and persistent inflammation driven by neutrophilic immune pathways. Patients living with GPP frequently experience cycles of remission and sudden flare-ups, which can be severe, debilitating, and potentially life-threatening.
The newly presented data focus on the long-term impact of subcutaneous (SC) SPEVIGO therapy on flare frequency. Preliminary results demonstrate a substantial reduction in disease activity among treated patients. Before entering the EFFISAYIL clinical program, patients reported an average of approximately 2.0 flares per year. With continued long-term SC treatment, this rate dropped dramatically to a cumulative mean of just 0.13 flares per year.
Notably, among the 118 patients who received three or more years of subcutaneous SPEVIGO treatment, nearly three-quarters—74.6% (88 patients)—experienced no flares at all during that period. This outcome represents a meaningful improvement in disease control, suggesting that long-term maintenance therapy with SPEVIGO may significantly reduce the burden of recurrent flares, which are a hallmark of GPP.
The importance of these findings lies in the chronic and unpredictable nature of GPP. Unlike more common dermatological conditions, GPP involves systemic inflammation and can lead to complications beyond the skin, including fever, fatigue, and organ involvement. As such, reducing flare frequency is a critical therapeutic goal, as it directly impacts patient quality of life, reduces healthcare utilization, and may lower the risk of severe complications.
Shannon Schneider, Vice President of North America Medical Affairs at LEO Pharma, emphasized the significance of the results, noting that the long-term data further strengthen the evidence base supporting SPEVIGO as an effective treatment option for GPP. She highlighted the company’s ongoing commitment to addressing the unmet needs of patients living with this rare and often overlooked condition through continued research and data generation.
In addition to the subcutaneous maintenance therapy findings, the study also evaluated the use of intravenous (IV) SPEVIGO for the treatment of acute flares. Among the 131 patients enrolled in the EFFISAYIL ON study, 21 patients (16.0%) required IV treatment to manage a total of 36 flare events. The results indicate that IV SPEVIGO is effective in rapidly controlling flare symptoms.
Clinical assessments using the GPP Physician Global Assessment (GPPGA) scoring system demonstrated that 50.0% of flare treatments achieved a pustulation subscore of 0 (indicating complete clearance of pustules) within one week of IV administration. This rate increased to 58.3% by Week 2. Similarly, a GPPGA total score of 0 or 1 (clear or almost clear skin) was achieved in 41.7% of treatments at Week 1 and improved to 55.6% by Week 2.
These results suggest that IV SPEVIGO provides rapid and meaningful clinical improvement during acute flare episodes, complementing the long-term disease control achieved with subcutaneous administration. Together, the two routes of administration support a comprehensive treatment strategy that addresses both the chronic and episodic aspects of GPP.
Safety and tolerability data from the study were consistent with the known safety profile of SPEVIGO. In the subcutaneous treatment group, adverse events leading to treatment discontinuation were reported in 4.2% of patients. In the intravenous treatment group, serious adverse events occurred in 36.6% of patients, while discontinuation due to adverse events was reported in 4.6% of cases. These findings indicate that the therapy remains generally well tolerated over long-term use, with manageable safety outcomes.
Arash Mostaghimi, Assistant Professor of Dermatology at Brigham & Women’s Hospital, commented on the broader implications of the data. He noted that the results highlight the complementary roles of intravenous and subcutaneous SPEVIGO in managing GPP over the long term. According to Mostaghimi, this dual approach enables clinicians to address both the chronic baseline disease and the acute flare episodes that define the patient experience in GPP.
The presentation of these findings at the AAD Annual Meeting is part of LEO Pharma’s broader scientific program, which includes 17 accepted abstracts spanning its dermatology portfolio and pipeline. This extensive presence underscores the company’s continued investment in advancing medical dermatology and addressing unmet needs across a range of skin conditions.
From a clinical perspective, the availability of long-term data is particularly valuable in rare diseases like GPP, where limited patient populations and variable disease courses can make it challenging to establish robust evidence. The EFFISAYIL ON study contributes important insights into the durability of treatment response, the prevention of disease flares, and the overall safety of long-term therapy.
The results also reflect a growing trend in dermatology toward targeted biologic therapies that address specific immune pathways involved in disease pathogenesis. SPEVIGO, which targets the interleukin-36 (IL-36) receptor pathway, represents a novel mechanism of action in the treatment of GPP. By inhibiting this pathway, the therapy aims to reduce inflammation and prevent the cascade of immune responses that lead to flare development.
For patients, the implications of these findings are significant. Reduced flare frequency, improved disease control, and effective management of acute episodes can translate into better quality of life, fewer hospitalizations, and greater confidence in managing a chronic condition. For clinicians, the availability of both maintenance and acute treatment options within a single therapeutic framework provides greater flexibility in tailoring care to individual patient needs.
Looking ahead, continued follow-up from the EFFISAYIL ON study will be critical in further characterizing the long-term benefits and safety of SPEVIGO. As more data become available, they may help inform clinical guidelines and support broader adoption of the therapy in the management of GPP.
In summary, the long-term results presented by LEO Pharma demonstrate that SPEVIGO offers sustained efficacy in reducing flare frequency, effective control of acute flares, and a consistent safety profile in patients with generalized pustular psoriasis. These findings reinforce the therapy’s role as a key treatment option in addressing the complex and unpredictable nature of this rare dermatological disease, while highlighting the importance of ongoing research and innovation in improving patient outcomes.
About EFFISAYIL ON Study
The primary objective of this ongoing open-label, multicenter, 5-year long-term extension of the EFFISAYIL 1 and EFFISAYIL 2 trials is to assess the long-term safety and efficacy of SPEVIGO® (spesolimab-sbzo) treatment in patients with GPP. Subjects received SC SPEVIGO (300 mg every 4, 6, or 12 weeks; dose escalation and de-escalation for flare recurrence/resolution allowed per protocol) for up to 252 weeks. In case of a protocol-defined GPP flare, patients received IV SPEVIGO 900 mg (≤2 doses within 1 week).
The primary outcome measure of the study was the occurrence of treatment-emergent adverse events (TEAEs) up to Week 252 of maintenance treatment. The secondary outcome measures the reoccurrence of a GPP flare (defined by the GPPGA), the time to first achievement of a GPPGA score of 0 or 1 in patients who received flare rescue treatment, a GPPGA pustulation sub-score of 0 indicating no visible pustules (by visit), and the change from baseline in Psoriasis Symptom Scale (PSS) score (by visit).1,2,5
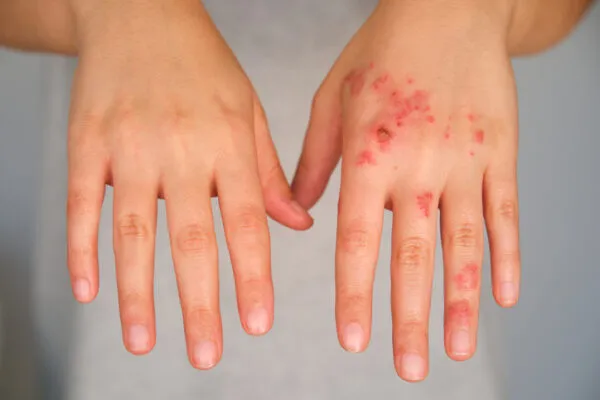
About Generalized Pustular Psoriasis
Generalized pustular psoriasis (GPP) is a chronic, heterogeneous, neutrophilic inflammatory disease associated with skin and systemic symptoms that is distinct from plaque psoriasis. GPP is recognized as a separate clinical entity from other forms of psoriasis, with the IL-36 pathway being a key driver of GPP and triggering response to treatment.3,4
GPP can become life-threatening (mortality rates ranging from 2% to 16%) due to severe complications, such as multisystem organ failure and sepsis requiring urgent hospital care; many GPP patients also suffer from various comorbidities, which contribute to the ongoing burden for the patient and healthcare systems.6,7 GPP symptoms appear unpredictably and present on a continuum, which greatly impacts a patient’s quality of life, and may cause fear and anxiety over the disease course, as well as long-term impacts on quality of life related to work/school, emotional health, social activities and finances.7,8
About SPEVIGO® (spesolimab-sbzo)
SPEVIGO® (spesolimab-sbzo)is a humanized, selective antibody that specifically blocks the activation of the IL-36R, a signaling pathway within the immune system shown to be involved in the pathogenesis of several autoinflammatory diseases, including GPP.9 It is the first targeted therapy for the treatment of GPP and has been evaluated in the largest clinical program specifically for the treatment of patients with GPP.10-12
Source Link:https://www.businesswire.com/