Zai Lab shares preclinical data showing ZL-1503 delivers rapid itch relief and reduces inflammation in atopic diseases
Zai Lab Limited has announced promising new preclinical findings for ZL-1503, its internally developed IL-13/IL-31Rα bispecific antibody, highlighting the therapy’s potential to address both inflammation and severe itch associated with atopic diseases. The data were presented in a scientific poster at the IMMUNOLOGY2026 conference held in Boston, Massachusetts, and provide early but compelling evidence supporting ZL-1503 as a potential first-in-class therapeutic candidate for moderate-to-severe atopic dermatitis and related conditions driven by IL-13 and IL-31 signaling pathways.
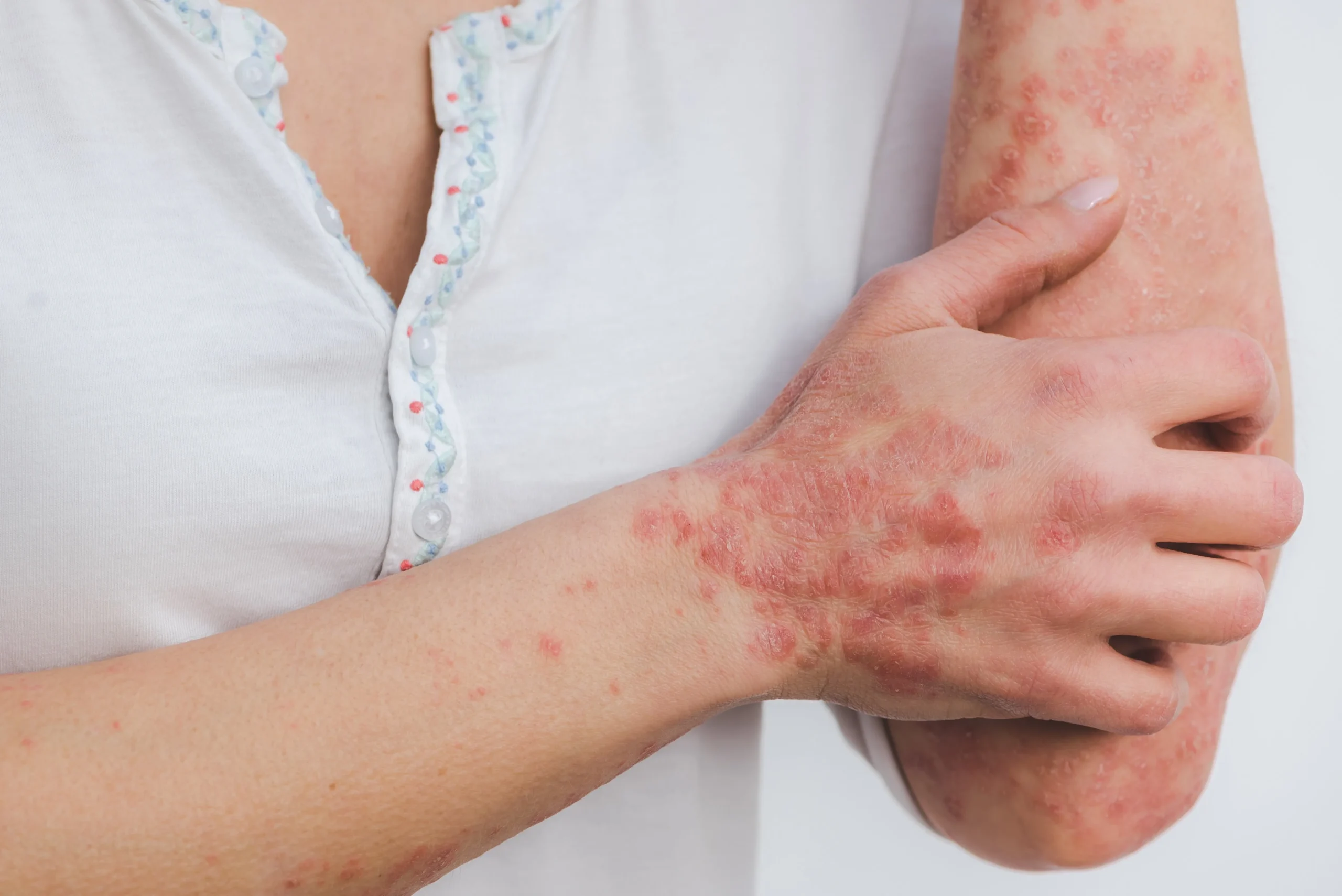
Atopic diseases, including atopic dermatitis, asthma, allergic rhinitis, and allergic conjunctivitis, are characterized by chronic inflammation and, in many cases, debilitating pruritus (itch). These conditions are often driven by dysregulated immune responses involving Type 2 helper T-cell (TH2) pathways. Among the key cytokines involved, IL-13 and IL-31 play central and complementary roles. IL-13 contributes to tissue inflammation, remodeling, and barrier dysfunction, while IL-31 is strongly associated with the sensation of itch and the amplification of local immune responses. Together, these cytokines sustain the so-called “itch-scratch-inflammation” cycle, in which scratching exacerbates inflammation, leading to further itching and disease progression.
Current therapeutic approaches often target individual cytokines or pathways, which may limit their effectiveness in fully controlling disease symptoms. ZL-1503 has been specifically engineered to overcome this limitation by simultaneously targeting both IL-13 and IL-31 receptor alpha (IL-31Rα), thereby disrupting two critical drivers of atopic disease in a single therapeutic intervention. This dual-targeting strategy is designed to provide more comprehensive and durable symptom relief compared to single-pathway inhibitors.
According to Rafael G. Amado, M.D., President and Head of Global Research and Development at Zai Lab, the breadth and durability of activity observed in the preclinical studies reinforce the company’s confidence in this approach. He emphasized that the ability of ZL-1503 to suppress both pruritus and inflammation across multiple disease models, particularly following a single dose, represents a significant advancement in the development of next-generation therapies for atopic conditions. He further noted that the findings support continued clinical development and suggest the therapy could meaningfully improve outcomes for patients suffering from these chronic and often burdensome diseases.
The preclinical study presented at IMMUNOLOGY2026 was designed to evaluate multiple aspects of ZL-1503’s biological activity, including its ability to inhibit IL-31-induced scratching behavior and IL-13-mediated signaling pathways. Researchers also assessed the therapy’s efficacy in several disease-relevant models, including asthma, allergic rhinitis, and allergic conjunctivitis, all of which were induced through controlled allergen challenges to mimic real-world disease mechanisms.
One of the most notable findings from the study was the durability of ZL-1503’s effects. A single intravenous dose, administered at varying levels of 0.3, 3, and 10 mg/kg, resulted in sustained and dose-dependent inhibition of both IL-31-induced scratching and IL-13-dependent pSTAT6 signaling. Remarkably, these effects were maintained over a 112-day observation period, indicating long-lasting suppression of the underlying disease pathways. This extended duration of action could translate into less frequent dosing in clinical settings, potentially improving patient adherence and quality of life.
In addition to reducing itch and molecular signaling associated with inflammation, ZL-1503 demonstrated significant therapeutic benefits across multiple organ systems affected by atopic disease. At the highest tested dose of 10 mg/kg, the therapy improved lung function and reduced airway inflammation in preclinical asthma models. It also alleviated symptoms associated with allergic rhinitis and conjunctivitis, suggesting broad applicability across a range of atopic conditions.
Further supporting these findings, transcriptomic and histopathological analyses revealed that ZL-1503 exerted a comprehensive impact on immune activity at the tissue level. The therapy was associated with suppression of TH2-related inflammatory pathways, reduced infiltration of immune cells, and decreased activation of mast cells in key tissues, including the lungs, nasal mucosa, and conjunctiva. These mechanistic insights provide a deeper understanding of how ZL-1503 modulates the immune system and reinforce its potential to address the root causes of atopic disease rather than merely alleviating symptoms.
Safety is a critical consideration in the development of any new therapeutic, particularly those targeting immune pathways. In this regard, ZL-1503 demonstrated a favorable nonclinical safety profile across all evaluated studies. No significant safety concerns were identified, supporting the continued advancement of the therapy into human clinical trials.
Building on these encouraging preclinical results, Zai Lab initiated Phase 1/1b first-in-human clinical studies of ZL-1503 in December 2025. These trials are currently underway in Australia, New Zealand, and China, and include both healthy volunteers and adult patients with moderate-to-severe atopic dermatitis. The studies are designed to evaluate the safety, tolerability, pharmacokinetics, and preliminary efficacy of the therapy, providing critical data to guide дальнейшее clinical development.
The advancement of ZL-1503 into clinical trials reflects a broader trend in immunology and dermatology toward more targeted and mechanism-based therapies. As understanding of the molecular drivers of atopic disease continues to evolve, there is growing recognition of the need for treatments that address multiple pathways simultaneously. By targeting both IL-13 and IL-31, ZL-1503 represents a novel approach that could potentially redefine the standard of care for patients with moderate-to-severe disease.
The burden of atopic dermatitis and related conditions extends beyond physical symptoms. Chronic itch, sleep disruption, and visible skin lesions can significantly impact mental health, daily functioning, and overall quality of life. For many patients, existing treatments provide only partial relief, underscoring the need for more effective and durable therapeutic options.
ZL-1503’s dual mechanism of action and sustained activity profile position it as a promising candidate to meet this need. If the results observed in preclinical studies are replicated in clinical trials, the therapy could offer a new level of symptom control and disease management for patients who currently have limited options.
The data presented at IMMUNOLOGY2026 also highlight the strength of Zai Lab’s research and development capabilities. By leveraging advanced antibody engineering and a deep understanding of immune biology, the company is building a pipeline of innovative therapies aimed at addressing complex diseases. ZL-1503, in particular, exemplifies this approach, combining scientific innovation with a clear focus on patient needs.
As the clinical development of ZL-1503 progresses, further studies will be essential to confirm its efficacy and safety in larger and more diverse patient populations. Researchers will also seek to better understand optimal dosing strategies, long-term outcomes, and potential applications beyond atopic dermatitis, including other IL-13 and IL-31-driven diseases.
In summary, the preclinical data presented by Zai Lab provide strong support for the continued development of ZL-1503 as a potential first-in-class bispecific antibody targeting IL-13 and IL-31 pathways. By addressing both inflammation and itch—the two central components of atopic disease—the therapy has the potential to break the cycle that drives disease progression and significantly improve patient outcomes. As it advances through clinical trials, ZL-1503 represents a promising step forward in the quest for more effective and durable treatments for atopic conditions.
About ZL-1503
ZL-1503 is a potential first-in-class bispecific antibody targeting IL-13 and IL-31Rα engineered by Zai Lab’s in-house discovery engine to simultaneously suppress both inflammatory and pruritogenic (itch-causing) pathways implicated in AD and other IL-13 and IL-31-driven diseases, potentially offering faster onset of action and superior efficacy compared to single-pathway inhibition.
ZL-1503 is currently being evaluated in the ongoing global Phase 1/1b clinical trial evaluating its safety, tolerability, pharmacokinetics, and efficacy. This clinical advancement is supported by preclinical studies in which ZL-1503 demonstrated the ability to simultaneously suppress inflammatory and pruritogenic pathways implicated in AD.
About Zai Lab
Zai Lab Limited (NASDAQ: ZLAB; HKEX: 9688) is an innovative, research-based, commercial-stage biopharmaceutical company based in China and the United States. We are focused on discovering, developing, and commercializing innovative products that address medical conditions with significant unmet needs in the areas of oncology, immunology, neuroscience, and infectious disease. Our goal is to leverage our competencies and resources to positively impact human health.
Source Link:https://www.businesswire.com/