LEO Pharma expands its psoriasis portfolio in China with NMPA approval of Enstilar®
LEO Pharma A/S has announced a significant regulatory milestone with the approval of Enstilar by China’s National Medical Products Administration (NMPA) for the treatment of adult patients living with plaque psoriasis. This development marks an important step forward in expanding access to innovative dermatological therapies in one of the world’s largest healthcare markets.
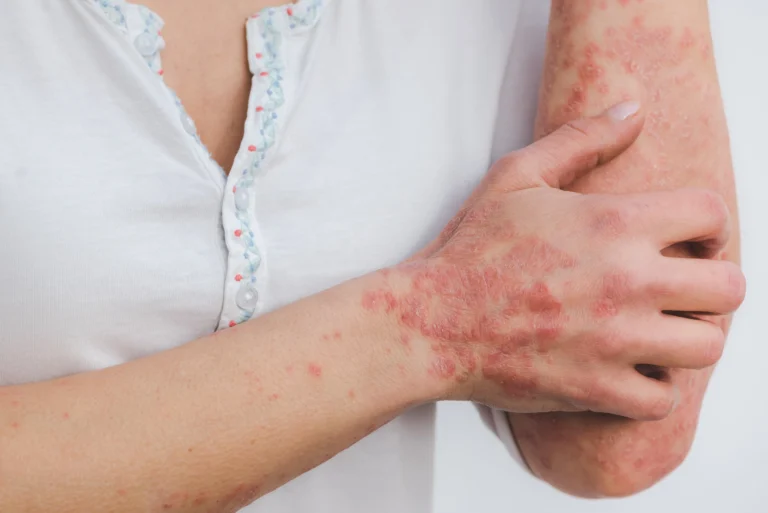
Plaque psoriasis, a chronic inflammatory skin condition characterized by red, scaly patches, affects an estimated 6.5 million people across China. Despite the availability of various treatment options, there remains a substantial unmet need for therapies that are both effective and convenient for long-term disease management. The approval of Enstilar introduces a new therapeutic option that has already gained widespread acceptance in international markets, offering hope for improved disease control and quality of life for millions of patients.
One of the distinguishing features of Enstilar is its foam-based formulation, which represents a meaningful advancement in topical treatment delivery. Unlike traditional ointments or creams, the foam is aerated, allowing for smoother and more uniform application across the skin. This is particularly beneficial for patients with lesions in hair-bearing areas, where conventional formulations can be difficult to apply effectively. The foam spreads easily, enhancing patient comfort and adherence to treatment, while also facilitating even distribution of its active ingredients—calcipotriene and betamethasone dipropionate. Clinical studies have demonstrated that this formulation can deliver rapid symptom relief, an important consideration for patients seeking quick improvements in itching, scaling, and inflammation.
From a strategic perspective, China represents a cornerstone of LEO Pharma’s long-term growth ambitions. The country’s large patient population, combined with a rapidly evolving healthcare infrastructure and increasing demand for innovative therapies, makes it a highly attractive market for global pharmaceutical companies. By securing NMPA approval for Enstilar, LEO Pharma not only strengthens its footprint in China but also reinforces its position as a global leader in medical dermatology.
Company leadership has emphasized the importance of this milestone. Executives have highlighted that China is the largest market globally in terms of psoriasis patient numbers and plays a critical role in the company’s international expansion strategy. The introduction of Enstilar is expected to complement LEO Pharma’s existing portfolio while also paving the way for future product launches in the region. The company continues to invest in research, development, and commercialization capabilities to support sustained growth and innovation in dermatology.
Globally, LEO Pharma has established itself as a market leader in topical psoriasis treatments, with nearly three million patients using its products worldwide. The addition of China to the list of more than 50 countries where Enstilar is approved further underscores the product’s global reach and clinical relevance. This widespread adoption reflects strong physician confidence in the therapy, as well as its demonstrated efficacy and safety profile across diverse patient populations.
The regulatory approval in China was supported by robust clinical evidence, including a Phase 3 study conducted in adult Chinese patients with stable plaque psoriasis. The trial enrolled 604 participants and compared the efficacy and safety of once-daily Enstilar foam with a commonly used ointment formulation containing the same active ingredients. After four weeks of treatment, Enstilar met both its primary and secondary endpoints, demonstrating superior outcomes compared to the ointment. These findings highlight the added value of the foam formulation in delivering improved therapeutic results.
Beyond clinical performance, the approval also signals progress in China’s broader healthcare landscape. In recent years, the country has placed increasing emphasis on accelerating access to innovative medicines, streamlining regulatory pathways, and improving patient outcomes. The availability of advanced treatments like Enstilar aligns with these goals, contributing to a more modern and patient-centered approach to disease management.
Local leadership at LEO Pharma China has described the approval as a key milestone in the company’s journey within the region. It not only expands treatment options for patients but also lays the groundwork for introducing a broader portfolio of dermatology therapies in the near future. The company aims to build a strong presence in China by combining global expertise with local insights, ensuring that its products meet the specific needs of Chinese patients and healthcare providers.
Looking ahead, LEO Pharma plans to launch Enstilar in China later this year, bringing the therapy to market and making it accessible to patients in need. The launch is expected to be supported by educational initiatives aimed at healthcare professionals, as well as efforts to raise awareness about psoriasis and its management. By fostering collaboration with the medical community and investing in patient support programs, the company seeks to maximize the impact of this new treatment option.
In summary, the NMPA approval of Enstilar represents a major advancement for both LEO Pharma and the millions of individuals living with plaque psoriasis in China. It combines clinical innovation with strategic market expansion, reinforcing the company’s commitment to improving dermatological care worldwide. As the therapy becomes available in China, it is poised to play a meaningful role in enhancing treatment outcomes and addressing the significant burden of psoriasis in the region.
About Enstilar®
Enstilar® is an aerosol spray foam containing calcipotriol monohydrate 50 mcg/g and betamethasone dipropionate 0.5 mg/g. In the EU, it is indicated for the treatment of psoriasis vulgaris in adults for up to 4 weeks. Patients who have responded at 4 weeks’ treatment using Enstilar once daily are suitable for long-term maintenance treatment. 4,6,7,8
About Psoriasis
Psoriasis is a chronic, systemic inflammatory disease that primarily affects the skin in 125 million people worldwide.9,10 Psoriasis is the result of skin barrier cell proliferation and the activation of cytokines (a family of proteins involved in immune responses) that cause inflammation.11 About 80% to 90% of patients are affected by plaque psoriasis, the most common clinical form of psoriasis.12 The symptoms of plaque psoriasis are itchy or painful raised scaly and inflamed plaques. Plaques may appear anywhere on the body, but often appear on the scalp, knees, elbows and torso.12
About LEO Pharma
LEO Pharma is a global leader in medical dermatology. We deliver innovative solutions for skin health, building on a century of experience with breakthrough medicines in healthcare. We are committed to making a fundamental difference in people’s lives, and our broad portfolio of treatments serves close to 100 million patients in over 70 countries annually. LEO Pharma is co-owned by majority shareholder the LEO Foundation and, since 2021, Nordic Capital. Headquartered in Denmark, LEO Pharma has a team of 4,000 people worldwide. Together, we reach far beyond the skin.
Source Link:https://www.businesswire.com/