
BioNTech Appoints Kylie Jimenez as Chief People Officer to Management Board
BioNTech Appoints Kylie Jimenez as Chief People Officer to Management Board BioNTech SE announced today that the Supervisory Board has appointed Kylie Jimenez to the Management Board as Chief People Officer…
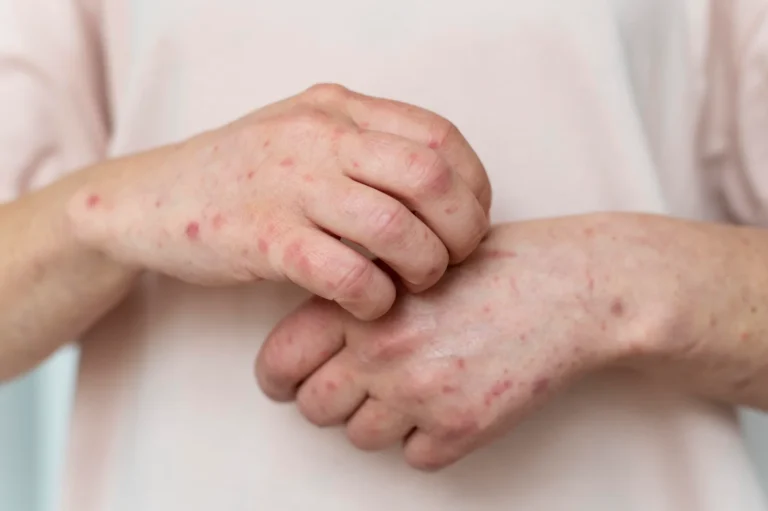
Litifilimab Earns FDA Breakthrough Designation for Cutaneous Lupus—First Targeted Therapy Hope
Biogen’s Litifilimab Earns FDA Breakthrough Therapy Designation for Cutaneous Lupus Erythematosus—First Targeted Therapy on the Horizon Biogen Inc. announced today that the U.S. Food and Drug Administration (FDA) has granted Breakthrough…

RCEM’s new film promotes a specialty to be proud of | AnnualConference 2026
This is the place for emergency medicine’ features real people working in emergency departments in Coventry and Manchester. Commissioned by the Royal College of Emergency Medicine (RCEM), the film is…
Intellia Therapeutics Announces FDA Lifts Clinical Hold on MAGNITUDE-2 Phase 3 Trial for ATTRv-PN
Intellia Therapeutics Announces FDA Lifts Clinical Hold on MAGNITUDE-2 Phase 3 Trial for ATTRv-PN Intellia Therapeutics, Inc., a leading clinical-stage gene editing company focused on revolutionizing medicine with CRISPR-based therapies,…
ICON’s Accellacare Site Network Enhances Oncology Research Capabilities Through New Cancer Institute Partnership
ICON’s Accellacare Site Network Enhances Oncology Research Through New Cancer Institute Partnership ICON plc today announced a significant expansion of oncology research capabilities within its Accellacare Site Network through the…

Will Forte Partners with Teva for “Honestly HD” Campaign to Raise Awareness About Huntington’s Disease
Will Forte Partners with Teva to Launch “Honestly HD”—A Candid Conversation About Huntington’s Disease Teva Pharmaceuticals, a U.S. affiliate of Teva Pharmaceutical Industries Ltd. (NYSE and TASE: TEVA), today announced…

DARZALEX FASPRO® Quadruplet Approved for Transplant-Ineligible Newly Diagnosed Multiple Myeloma
DARZALEX FASPRO® quadruplet approved in U.S. for transplant-ineligible newly diagnosed multiple myeloma Johnson & Johnson announced today the U.S. Food and Drug Administration (FDA) approved DARZALEX FASPRO® (daratumumab and hyaluronidase-fihj) in combination…

Neurocrine Biosciences Launches Phase 2 Trial of NBI-1065890 for Tardive Dyskinesia in Adults
Neurocrine Biosciences Launches Phase 2 Trial of NBI-1065890 for Tardive Dyskinesia in Adults Neurocrine Biosciences, Inc. today announced the initiation of its Phase 2 clinical study of investigational compound NBI-1065890…

BioMarin Announces Proposed Private Offering of Senior Notes and Launch of New Senior Secured Term Loan Facility
BioMarin Moves to Strengthen BalanceBioMarin Announces Proposed Private Offering of Senior Notes and New Senior Secured Term Loan Syndication BioMarin Pharmaceutical Inc. announced today that it intends to offer, subject…

WuXi Biologics, HanchorBio Partner on Next-Gen Fusion Proteins
WuXi Biologics and HanchorBio Forge Strategic Partnership to Advance Next-Gen Bi- and Multi-Functional Fusion Proteins WuXi Biologics, a global leading Contract Research, Development and Manufacturing Organization (CRDMO), and HanchorBio Inc.…

Bayer to Unveil New Phase III Data and Radiology Portfolio Innovations at ECR 2026
Bayer to Showcase New Phase III Data and Innovations in Radiology Portfolio at ECR 2026 Bayer will present advancements of its comprehensive radiology portfolio at the 2026 European Congress of…

GSK’s RSV Vaccine, Arexvy, Gains European Approval for Use in All Adults Aged 18 and Older
GSK’s RSV Vaccine, Arexvy, Gains European Approval for Use in All Adults Aged 18 and Older GSK plc today announced that its adjuvanted recombinant respiratory syncytial virus (RSV) vaccine, Arexvy,…

