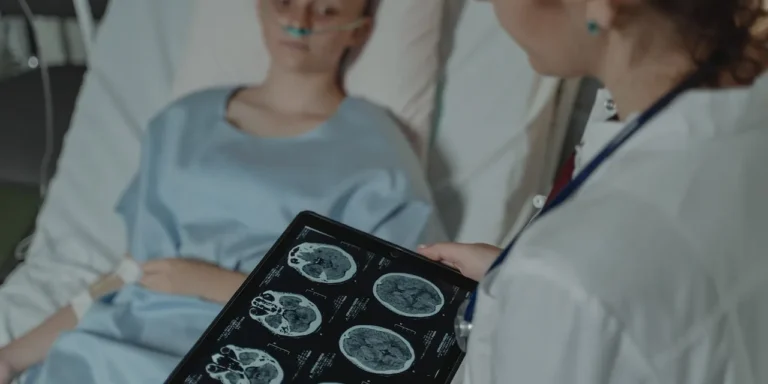
Researchers leverage cell self-destruction to treat brain tumors
Glioblastoma is the most common type of brain tumor in adults. The disease is 100% fatal and there are no cures, making it the most aggressive type of cancer. Such…

Glioblastoma is the most common type of brain tumor in adults. The disease is 100% fatal and there are no cures, making it the most aggressive type of cancer. Such…
CycPeptMPDB, a novel database – created by Tokyo Tech researchers – focused on the membrane permeability of cyclic peptides, could accelerate the development of drugs based on these promising compounds.…

How to determine the most effective treatment for colon cancer? The response to chemotherapy varies greatly from one patient to another. A team from the UNIGE has developed a new…

Pfizer Invites Shareholders to Attend Virtual-Only 2023 Annual Meeting of Shareholders on April 27 Pfizer Inc. (NYSE: PFE) announced today that its shareholders and the general public are invited to…

FDA Accepts Pfizer’s Supplemental New Drug Applications for BRAFTOVI + MEKTOVI Pfizer Inc. (NYSE:PFE) announced today that the U.S. Food and Drug Administration (FDA) has accepted for review the Supplemental…

GSK reaches agreement to acquire late-stage biopharmaceutical company BELLUS Health GSK plc (LSE/NYSE: GSK) and BELLUS Health Inc. (TSX/NASDAQ: BLU) today announced that they have entered into an agreement under…
Today, the U.S. Food and Drug Administration approved Omisirge (omidubicel-onlv), a substantially modified allogeneic (donor) cord blood-based cell therapy to quicken the recovery of neutrophils (a subset of white blood…

Gepotidacin’s positive phase III data shows potential to be the first in a new class of oral antibiotics for uncomplicated urinary tract infections in over 20 years GSK plc (LSE/NYSE:…

Today, the U.S. Food and Drug Administration approved Narcan, 4 milligram (mg) naloxone hydrochloride nasal spray for over-the-counter (OTC), nonprescription, use – the first naloxone product approved for use without…

Moderna and Merck Announce mRNA-4157 (V940), an Investigational Individualized Neoantigen Therapy, in Combination With KEYTRUDA® (pembrolizumab), Demonstrated Superior Recurrence-Free Survival in Patients With High-Risk Stage III/IV Melanoma Following Complete Resection…

Merck’s KEYTRUDA® (pembrolizumab) Plus Chemotherapy Significantly Improved Overall Survival Compared to Chemotherapy Alone in Patients With Advanced or Unresectable Biliary Tract Cancer RAHWAY, N.J.–(BUSINESS WIRE)– Merck (NYSE: MRK), known as MSD…
Merck Strengthens Immunology Pipeline with Acquisition of Prometheus Biosciences, Inc. RAHWAY, N.J. & SAN DIEGO–(BUSINESS WIRE)– Merck (NYSE: MRK), known as MSD outside the United States and Canada, and Prometheus Biosciences,…